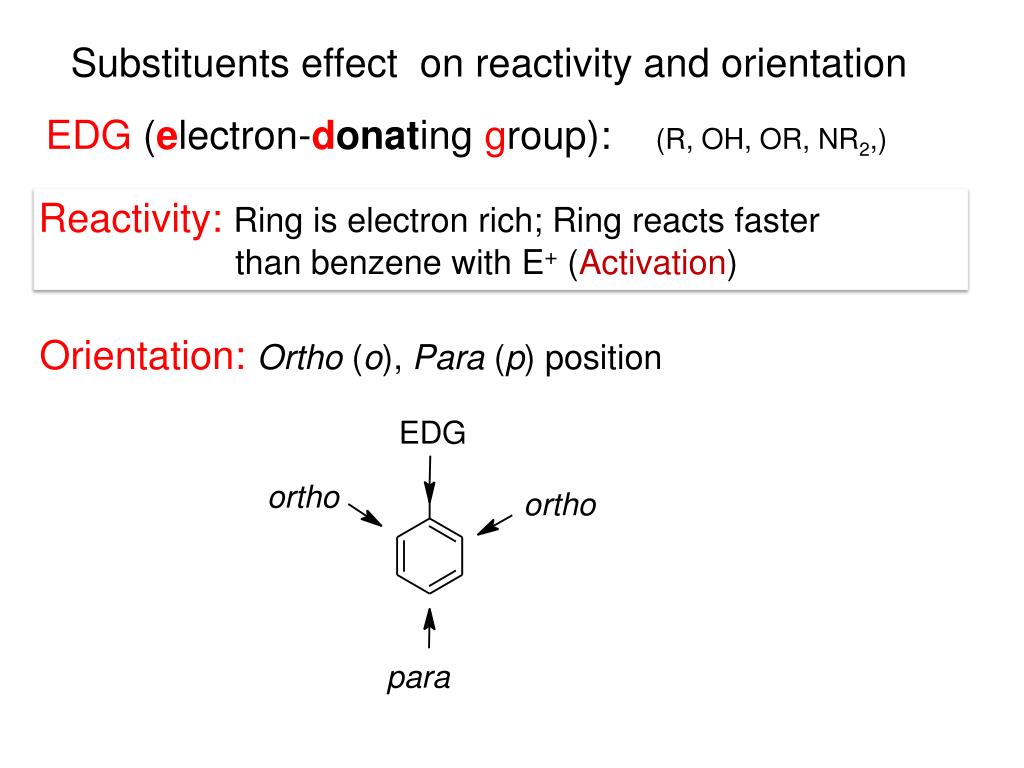
For aryl halides having this special kind of structure, nucleophilic substitution proceeds readily and can be used for synthetic purposes. However, aryl halides do undergo nucleophilic substitution readily Jf the aromatic ring contains, in addition to halogen, certain other properly placed groups: electron-withdrawing groups like NO2, NO, or CN, located ortho or para to halogen. We cannot use aryl halides as we use alkyl halides in the Friedel-Crafts reaction. Except for certain industrial processes where very severe conditions are feasible, one does not ordinarily prepare phenols ( ArOH), ethers (ArOR), amines (ArNH2), or nitriles (ArCN) by nucleophilic attack on aryl halides. It is typical ofvry\ halides that they undergo nucleophilic substitution only with extreme difficulty. (These reactions of diazonium salts have been discussed in detail in Sees. From the standpoint of synthesis, then, the nitro compounds hear much the same relationship to aryl halides that alcohols do to alkyl halides. Instead, aryl halides are most commonly prepared by replacement of the nitrogen of a diazonium salt as the sequence above shows, this ultimately comes from a nitro group which was itself introduced directly into the ring. 11.7) as the Lewis acid, one can direct bromination exclusively to the para position, (b) Alkyl halides are most often prepared from the corresponding alcohols aryl halides are not prepared from the phenols. Furthermore, by use of bulky thallium acetate (Sec. (a) Direct halogenation of the aromatic ring is more useful than direct halogenation of alkanes although mixtures may be obtained (e.g., ortho para], attack is not nearly so random as in the free-radical halogenation of aliphatic hydrocarbons. These methods, we notice, differ considerably from the methods of preparing alkyl halides. Chlorobenzene and bromobenzene, for example, have boiling points very nearly the same as those of /i-hexyl chloride and w-hexyl bromide like the alkyl halides, the aryl halides are insoluble in water and soluble in organic solvents. Unless modified by the presence of some other functional group, the physical properties of the aryl halides are much like those of the corresponding alkyl halides. 478) has named the base-catalyzed halogen dance (Problem 23, p. But we should be aware that there are others: free-radical reactions, for example, and what Joseph Bunnett (p. Of these reactions, we shall have time to take up only two. 
3.18), it is only necessary to provide the proper conditions and to have the ingenuity to observe what is going on. It is becoming increasingly evident that aryl halides are actually capable of doing many different things as with the "unreactive" alkanes (Sec. Before 1953, aryl halides appeared to undergo essentially only one reaction and that one, rather poorly. We must keep in mind that aryl halides are of *'low reactivity" only with respect to certain"sets of familiar reactions typical of the more widely studied alkyl halides. Moreover, this low reactivity is caused partly, at least by the same structural feature that is responsible for their anomalous influence on electrophilic attack: partial double-bond character of the carbon-halogen The parallel between aryl and vinyl halides goes further: both are unreactive toward nucleophilic substitution and, as we shall see, for basically the same reason. Problem 25.1 Drawing all pertinent structures, account in detail for the fact that: (a) nitration of chlorobenzene is slower than that of benzene, yet occurs predominantly ortho,para\ (b) addition of hydrogen iodide to vinyl chloride is slower than to ethylene, yet yields predominantly 1-chloro-l-iodoethane. Through its resonance effect, halogen releases electrons and tends to activate but only toward attack at certain positions. Through its inductive effect, halogen withdraws electrons and deactivates the entire molecule toward electrophilic attack. In both cases we attributed the influence of halogen to the working of opposing factors. 11.21) in electrophilic addition, halogen deactivates, yet causes Markovnikov orientation (Problem 11.13, p. In electrophilic substitution, halogen deactivates, yet directs ortho,para (Sec. In each of these reactions, halogen exerts an anomalous influence on reactivity and orientation. Each kind of compound contains another functional group besides halogen: aryl halides contain a ring, which undergoes electrophilic substitution vinyl halides contain a carbon-carbon double bond, which undergoes electrophilic addition. 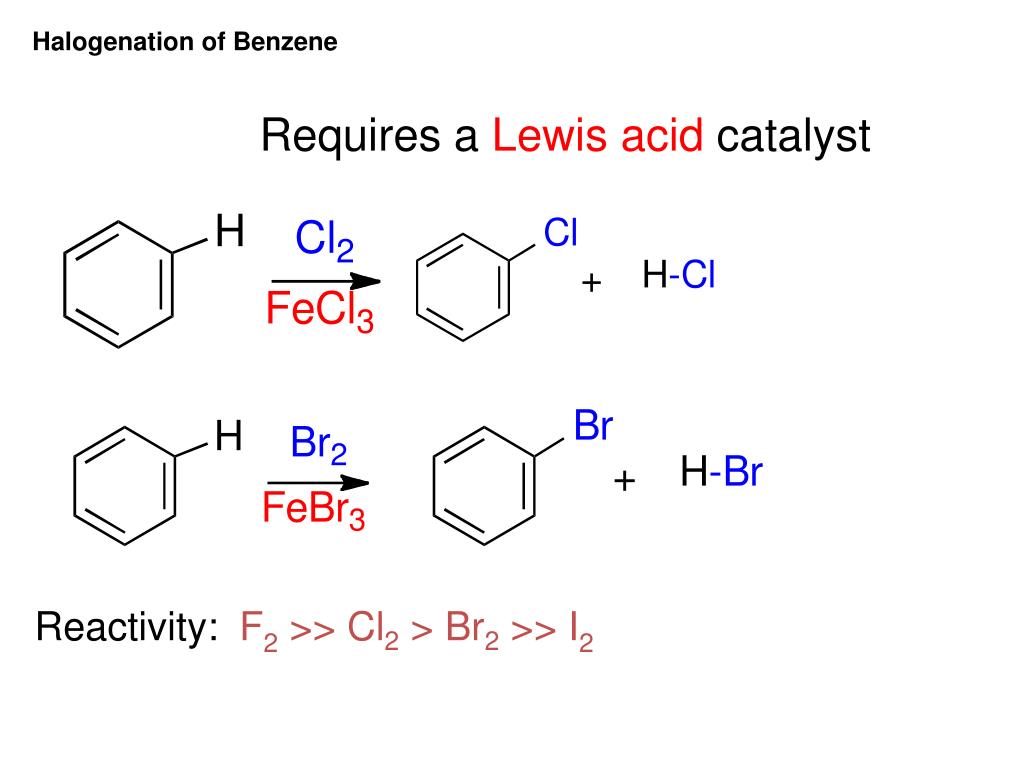
Vinyl halides, we have already seen, show an interesting parallel to aryl halides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
